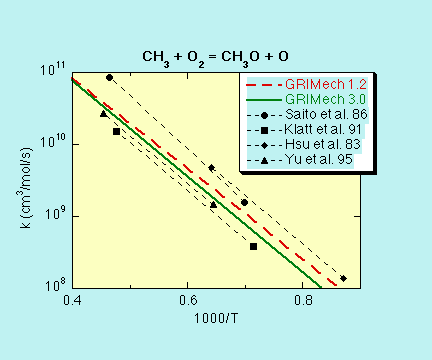
CH3 + O2 -> O + CH3O
k = 3.56E13 exp(-30480 cal/mol /RT) cm3/mol s
- SOURCE:
- Yu, C.-L., Wang, C., and Frenklach, M. (1995) J. Phys. Chem.
99, 14377.
- COMMENTS:
- The initial rate coefficient for GRI-Mech 1.1, 1.2, and 2.11 was a
consensus expression based upon results reported by
Saito, K., Ito, R., Kakumoto, T., and Imamura, A. (1986) J. Phys.
Chem. 90, 1422.
Klatt, M., Rohrig, M., and Wagner, H.Gg. (1991) Ber. Bunsenges.
Phys. Chem. 95, 1163.
Hsu, D.S.Y., Shaub, W.M., Creamer, T., Gutman, D., and Lin, M.C. (1983)
Ber. Bunsenges. Phys. Chem. 87, 909.
Yu, C.-L., Wang, C., and Frenklach, M. (1995) J. Phys. Chem.
99, 14377.
The rate coefficient then served as an optimization variable in GRI-Mech
1.1, 1.2, and 2.1 releases. As a result of the 1.2 optimization round,
the preexponential factor was lowered by 11% from the initially assigned
expression, and remained the same after the 2.1 optimization round.
The significantly lower rate coefficient expression from Yu et al. (1995)
was used for the
starting mechanism of GRI-Mech 3.0, and served as an optimization variable.
It was
multiplied by a factor of 1.24 as a results of the optimization.
______________________________________________________________________
Temp delta-S delta-H kf kr Keq
(K) (cal/mol K) (kcal/mol) ----(mol,cm3,s)-----
______________________________________________________________________
300 -2.3 28.3 2.22E-09 3.13E+12 7.11E-22
500 -3.0 28.1 1.69E+00 1.44E+13 1.18E-13
1000 -2.8 28.3 7.77E+06 4.76E+13 1.63E-07
1500 -2.2 29.0 1.29E+09 6.55E+13 1.97E-05
2000 -1.8 29.7 1.66E+10 7.23E+13 2.30E-04
2500 -1.4 30.6 7.71E+10 7.34E+13 1.05E-03
3000 -1.1 31.3 2.14E+11 7.24E+13 2.96E-03
_____________________________________________________________________
[ [ Rate coefficient format |
Chemkin file |
Thermo data |
Main menu ]
