REFERENCES
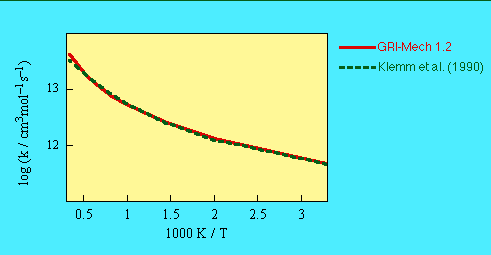
Total rate constant for k(25) + k(285) is the same as the GRI-Mech 1.2 value.
______________________________________________________________________
Temp delta-S delta-H kf kr Keq
(K) (cal/mol K) (kcal/mol) ----(mol,cm3,s)-----
______________________________________________________________________
300 9.1 -27.0 2.94E+11 7.09E-11 4.15E+21
500 9.6 -26.8 8.71E+11 1.38E+00 6.28E+13
1000 8.9 -27.4 3.45E+12 4.19E+04 8.24E+07
1500 8.0 -28.4 7.54E+12 9.69E+06 7.77E+05
2000 7.3 -29.7 1.30E+13 1.90E+08 6.82E+04
2500 6.7 -31.0 1.98E+13 1.33E+09 1.48E+04
3000 6.2 -32.4 2.78E+13 5.41E+09 5.12E+03
______________________________________________________________________